How many times do I have to tell you? Shut the refrigerator door!

Congratulations! You just bought a fancy new refrigerator. After you bring it home, you open the door and turn it on.
What will happen to the temperature of the room?
This section requires Javascript.
You are seeing this because something didn't load right. We suggest you, (a) try
refreshing the page, (b) enabling javascript if it is disabled on your browser and,
finally, (c)
loading the
non-javascript version of this page
. We're sorry about the hassle.
9 solutions
Do you also need to assume that the room is warmer than the refrigerator before to door is opened?
wow , Nicely Explained :)
perfectly explained
Why is that the heat rejected to room is more than the heat absorbed inside the refrigerator? What's the source of this additional energy? Please explain the same using the laws of thermodynamics.
This doesn't address the electrical input, save for the image. The explanation completely misses what causes the answer to be what the answer is.
The electric bill rises.
The temperature rises because heat is taken from the room and when the same heated is rejected, some further work done by the compressor is also added.So the heat rejected is greater.And then it will take heat from a more hotter room and so the temperature will go on increasing.
this problem is an isolated system with a single input from outside, ie, the electrical energy provided to the refrigerator. Using the First Law of Thermodynamics, the internal energy change in a system is the sum of all the inputs and outputs... as we have a net input, the temperate, which is a measure of the energy content of the room, will increase
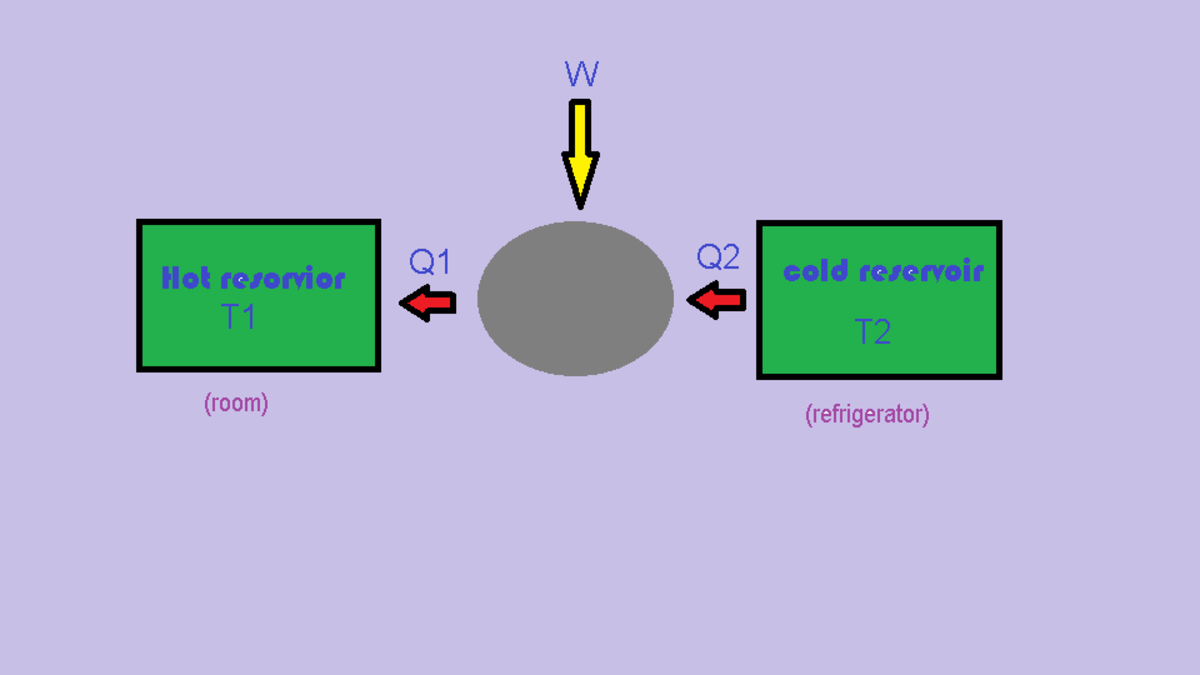
The room gets warm by opening the door of an electrical refrigerator.A refrigerator extracts heat from the freezing chamber(cold reservoir),some work is done on it by electric motor and rejects heat into the surrounding(hot reservoir) air. If the door of a running refrigerator is left open,more heat is rejected to the surrounding. Hence room will get slightly heated.
working of refrigerator is opposite to that of heat engine.
What is about the cooling of the refrigerator?
Since this is a closed system and we are putting energy into it by turning the refrigerator on (through the electrical wiring), the temperature should go up.
but i have question rise of temp in refrigerator means wat is the is cooling is increased or heat is increased
The average temperature of the room and refrigerator will increase (Second Law of Thermodynamics). Although the region close to the evaporator (inside the fridge) will get cooler, heat removed from the air around the evaporator is transferred to the condenser and from there to the air in the room. In addition to this, the work done to transfer heat from the interior to the exterior of the refrigerator - i.e. the operation of the compressor in an electrical refrigerator - adds heat to the room. This of course assumes no heat loss from the room and also that the evaporator is warm enough for the refrigerant to vaporize.
The (assuming it's a room where the heat cannot escape) magically cools down, because everyone knows that you can use energy to reduce heat, thus losing energy and violating physics.
The Question is easily solved according to the principles of thermodynamics. A we know that refrigerator has to do a sum of work on the system to coll it and gives out heat Q2 which is equal to the sum of the heat absorbed from the internal system and the work done by the Carnot engine . thus when we open the door each time there adds up a sum of energy in the room and the "TEMPERATURE RISES".
EASY ISN'T IT.
Firstly, lets see how a normal refrigerator works (when door is closed): Refrigerators work as heat pumps (but reverse). With the help of a compressor they absorb some heat from inside the refrigerator compartment and reject some amount of heat to the room (via condenser). In other words the heat rejected to room is more than the heat absorbed inside the refrigerator.
Now consider the case when door is open: As the door is open the heat Q 2 is absorbed from the entire room instead of just the refrigerator compartment. While the heat Q 1 is rejected to the room just as before. Q 1 > Q 2 That is, the heat rejected is more than heat absorbed in the room. Hence there is a net heat addition in the room and the temperature of room would increase
→ We are assuming for the sake of simplicity that: 1 . There are no other energy interactions going on in the room, i.e. no heater, air conditioner or a stove is being operated in the room. 2 . The walls of the room are adiabatic, i.e the outside weather does not affect the room conditions. 3 . The compressor is 100% efficient with no irreversible losses in the form of heat.